Malic acid



pictures of structural formula & malic acid
Basic informations about malic acid:
Generally, marlic acid is an organic acid and also a compound.
It has a chemical formula of HO2CCH2CHOHCO2H . Which has
a ph value of around 3-5.
Malic acid was first isolated from apple juice by Carl Wilhelm Scheele in 1785.Antoine Lavoisier in 1787 proposed the name acide malique which is derived from the Latin word for apple, mālum.[
Facts about malic acid:
Do you realise that malic acid is used in variety of food?
Some examples are : chewing gums, fruits, deserts, bakery
products, bevarages--- both carbonated & carbonated, hard/
soft candies and last for not least skin & medical care products.



However why is it so? Why is it used in so many variety and types of products?
The reason of malic acid used in variety of products is due to its properties.
Malic acid has:
- a clean, mellow, smooth, persistent sourness,
- flavour enhancement and blending abilities,
- a high solubility rate,
- lower hygroscopicity than Citric or Tartaric acids,
- a lower melting point than other acids for easier incorporation into molten confections,
- and good chelating properties with metal ions.
It forms:
- economical acidulant blends with other acids,
- more soluble calcium salts than Citric acid, and
- effective buffering mixtures.
-Olso it can help to improve Ph stability which is it's unqiue feature.
This is how they are applied to the examples and features of malic acid products I mentioned above:
Hard Candy
Malic acid boosts sourness intensity and enhances fruit flavours. It has a lower melting point than other food acids - this means that it can be incorporated into the molten hard candy without added water - shelf life is increased since the initial moisture level in the hard candy is lower.
Soft Candy
In agar, gelatin or pectin-based candies such as jellies and gummies, Malic acid is used to achieve a natural fruit flavour profile, proper gelling and good product clarity.
Chewing Gum
Organic food acids combined with saccharin improve saliva stimulation in chewing gum. Malic acid is preferred due to its flavour enhancement properties. Using blends of acids with different partition coefficients results in a sequential release of acid - this creates prolonged juiciness and flavour during chewing.

Acid-Based Facial Products
Malic acid, an alpha hydroxy fruit acid, can be used in skin care products to rejuvenate and improve skin conditions.

BAKERY PRODUCTS
Bakery products with fruit fillings (cookies, snack bars, pies, and cakes) have a stronger and more naturally balanced fruit flavour when the fruit filling includes Malic acid. Pectin gel texture is more consistent due to Malic acid's buffering capacity.

MEDICAL AND PERSONAL CARE PRODUCTS
In throat lozenges, cough syrups, and effervescent powdered preparations, Malic acid enhances fruit flavour and can diminish the flavour impact of active components. As Malic acid stimulates saliva flow, it can be used in tooth-cleaning preparations and mouthwashes. Germicidal compounds are used in combination with Malic acid in soaps, mouthwashes, and toothpaste.

BEVERAGES
Carbonated Beverages
Adding Malic acid improves economies, especially in artificially sweetened products. Flavours are enhanced, allowing less flavour to be used, and the overall flavour profile is broader and more natural.
Non-carbonated Beverages
Malic acid is a preferred acidulant for still beverages (fruit drinks, nectars, iced-teas, sports drinks, calcium fortified juices), because it enhances fruit flavours, improves pH stability, and masks the aftertaste of some salts.
Powdered Mixes
In iced tea, sports drink or fruit soup dry mixes, Malic acid is preferred due to its rapid dissolution rate and flavour enhancement qualities. Since Malic acid provides more sourness than Citric acid, less acidulant is required and unit weight can be reduced.

Low Calorie Beverages
In beverages containing intense sweeteners, less Malic acid than Citric is required to achieve the desired sourness and flavour at a higher pH. Malic acid's extended sourness masks sweetener aftertaste (see Taste retention chart) and its blending and fixative abilities give a balanced taste. In a study with 14-30 year olds, aspartame sweetened low-calorie soft drinks acidified with Malic acid were preferred over those with Citric acid.
ESSENTIAL METABOLISM
Malic acid is formed in metabolic cycles in the cells of plants and animals, including humans. For instance, in both the KREB and glyoxalate cycles it provides the cells with energy and carbon skeletons for the formation of amino acids. A relatively large amount of Malic acid is produced and broken down in the human body every day.
Chromic acid

The term chromic acid is usually used for a mixture made by adding concentrated sulfuric acid to a dichromate, which may contain a variety of compounds, including solid chromium trioxide.
Uses
Chromic acid is an intermediate in chromium plating, and is also used in ceramic glazes, and colored glass. Because a solution of chromic acid in sulfuric acid (also known as a sulfochromic mixture orchromosulfuric acid) is a powerful oxidizing agent, it can be used to clean laboratory glassware, particularly of otherwise insoluble organic residues. This application has declined due to environmental concerns
Hexavalent chromium compounds are toxic and carcinogenic. For this reason, chromic acid oxidation is not used on an industrial scale.
Fumaric acid


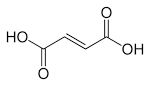
pictures of structural formula & fumaric acid
Basic information about fumaric acid:
Generally, fumaric acid is an organic acid and also a compound which is widely
found in nature. In humans and other mammals, Fumaric acid is a key intermediate in the tricarboxylic acid cycle for organic acid biosynthesis (the KREBS cycle). Fumaric acid is also an essential ingredient in plant life. It has a chemical formula of HO2CCH=CHCO2H . Which has a ph value of around 3 - 4.5 and also Fumaric acid was first prepared from succinic acid.[5] A traditional synthesis involves oxidation of furfural (from the processing of maize) using chlorate in the presence of a vanadium-based catalyst
Facts about fumaric acid:
It's common point with malic acid is that fumaric acid is also used
in our daily lifes for example--- variety of food , berverages,bakery
products and deserts.
However, it has other uses different from malic acid example it is used
in jams and jellies, egg white foams, pie fillings,cleaning agents for
dentures/bath salts and animal feed.
Also fumaric acid is the strongest organic food acid in titratable acidity and in sourness of course in comparison with malic acid fumaric acid is much stronger in sourness.



However why is fumaric acid accomodated to these products?
Fumaric acid has:
Long Lasting Sour Taste at pHs less than 4.5
Fumaric acid's hydrophobic nature results in persistent, long lasting sourness and flavour impact.
Lower pH with Minimal Sourness at pHs greater than 4.5
In food and beverage products with a pH greater than 4.5, Fumaric acid decreases the pH with minimal added sourness.
Greater Buffering Capacity at pH 3.0
Fumaric Acid has more buffering capacity than other food acids at pHs near 3.0 - this is due to its pKa1 and to its low molecular weight.
Anti-microbial Effects at Low pHs
Fumaric acid lowers pH in foods and beverages - this enhances the effectiveness of anti-microbial ingredients. In addition, undissociated Fumaric Acid that is present at low pHs has a bactericidal effect against E.Coli O157:H7.
Improves Dough Machinability
Fumaric acid blocks or breaks gluten disulphide bonds - this improves dough machinability in wheat flour doughs.
Delayed Action Leavening Acid
Granular Fumaric acid dissolves during baking and generates carbon dioxide by reaction with sodium bicarbonate. Fumaric acid has a higher neutralisation value than other delayed action leavening acids.
Non-hygroscopic
Fumaric acid absorbs virtually no atmospheric moisture - it stays dry and free flowing. Dry-mix products will not cake or harden, even during high humidity storage conditions, when Fumaric acid is used as the acidulant.
Better Flavour Stability
Fumaric acid does not absorb moisture that degrades flavour ingredients in dry mix products.
Lower Ingredient Cost
Ingredient cost is reduced when using Fumaric acid for two reasons. Fumaric acid is stronger than all other organic food acids in terms of sourness, acid equivalent weight and pH values - therefore, less can be used. In addition, Fumaric acid is more economical than other food acids on a cost per unit weight basis.
Lower Packaging Material Cost for Dry Mixes
For dry mixes, packaging material cost is decreased due to reduced moisture barrier requirements and because a smaller package can be used (the unit weight of the dry mix package can be lowered because less acidulant is used).
This is how they are applied and used to the examples and features i meantion about fumarlic acid.
Egg White Foams
Fumaric acid can promote maximum volume in both egg-white foams and end products based on egg-white foams. Fumaric acid can replace the more expensive cream of tartar to control egg-white volume. With Fumaric acid, egg whites can be overbeaten for as much as double the customary optimum time. Well suited for continuous flow processes, Fumaric acid can be added to both liquid and dried egg whites.


Cleaning Agents for Dentures/Bath Salts
The carbon dioxide generating compounds containing NaHCO3, K2CO3 and powdered Fumaric acid can be tableted with other ingredients to make cleaning agents for dentures and bath salts.


Animal Feed
Fumaric acid has proven to be a particularly effective additive to piglet feed during the post-weaning period. The inclusion of Fumaric acid and the resultant adjustment of the pH value demonstrate improved weight gain, food consumption and feed conversion ratio.


Pie Fillings
In pie fillings, Fumaric acid can be mixed directly with the starch and sugar ingredients as it is non-hygroscopic. Fumaric acid lowers costs by reducing the quantity of food acid needed in product formulations. Fumaric acid also improves smoothness and extends the critical cook times for optimum gelation

Jellies and Jams
Fumaric acid can cut food acid costs when used as an acidulant for jams, jellies and preserves. As little as two pounds of Fumaric acid can be used to replace every three pounds of Citric, Malic or Tartaric acid. At the 2:3 replacement ratio, Fumaric acid does not produce significant differences in gel strength or pH.
Comments (0)
You don't have permission to comment on this page.